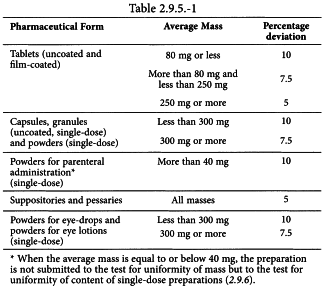
Uniformity of dosage units—comparative study of methods and specifications between Eur. Pharm. 3rd and USP 23 - ScienceDirect

PDF) Evaluation of the Discrepancy between the European Pharmacopoeia Test and an Adopted United States Pharmacopoeia Test Regarding the Weight Uniformity of Scored Tablet Halves: Is Harmonization Required? | Abdel Zaid -
![PDF] Weight and content uniformity of lorazepam half-tablets: A study of correlation of a low drug content product. | Semantic Scholar PDF] Weight and content uniformity of lorazepam half-tablets: A study of correlation of a low drug content product. | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/3f129f060436923435cbb5e514e797c1b6799cb1/3-Table2-1.png)
PDF] Weight and content uniformity of lorazepam half-tablets: A study of correlation of a low drug content product. | Semantic Scholar

ASTM E2810-19 - Standard Practice for Demonstrating Capability to Comply with the Test for Uniformity of Dosage Units
Updates of Ph. Eur. dosage form monographs and general chapters – Users invited to comment in Pharmeuropa 33.1 - European Directorate for the Quality of Medicines & HealthCare
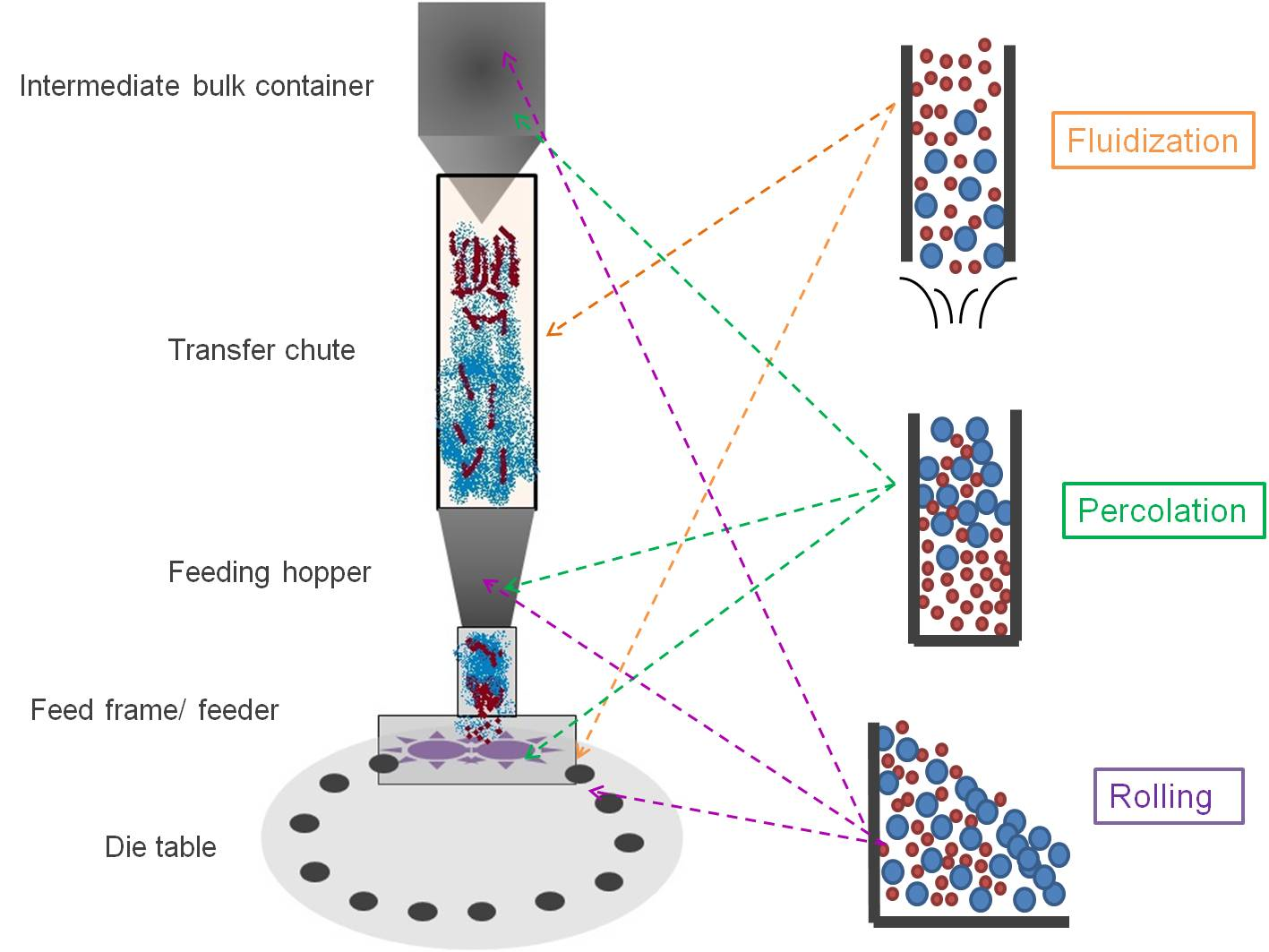
Pharmaceutics | Free Full-Text | Blend Segregation in Tablets Manufacturing and Its Effect on Drug Content Uniformity—A Review

Influence of Process Parameters on Content Uniformity of a Low Dose Active Pharmaceutical Ingredient in a Tablet Formulation According to GMP – topic of research paper in Chemical sciences. Download scholarly article

Uniformity of dosage units—comparative study of methods and specifications between Eur. Pharm. 3rd and USP 23 - ScienceDirect
![PDF] Weight and content uniformity of lorazepam half-tablets: A study of correlation of a low drug content product. | Semantic Scholar PDF] Weight and content uniformity of lorazepam half-tablets: A study of correlation of a low drug content product. | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/3f129f060436923435cbb5e514e797c1b6799cb1/3-Table3-1.png)